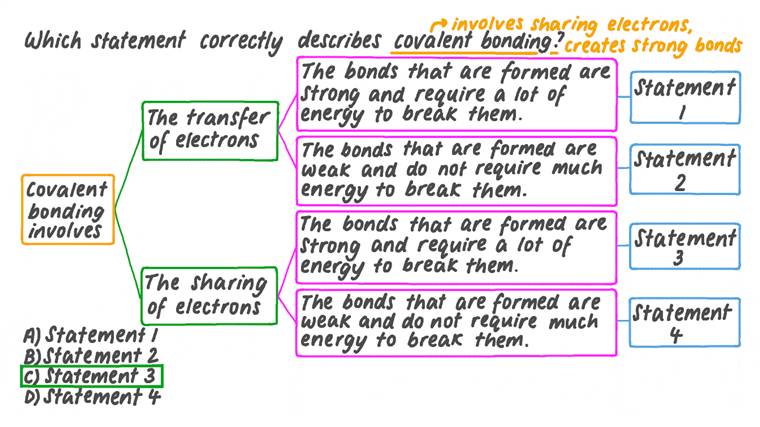
Lesson: Covalent Bonds Science
In this lesson, we will learn how to describe the formation of covalent bonds in simple molecules.
Lesson Plan
Lesson Video
Lesson Playlist
-
 +4
01:32
+4
01:32